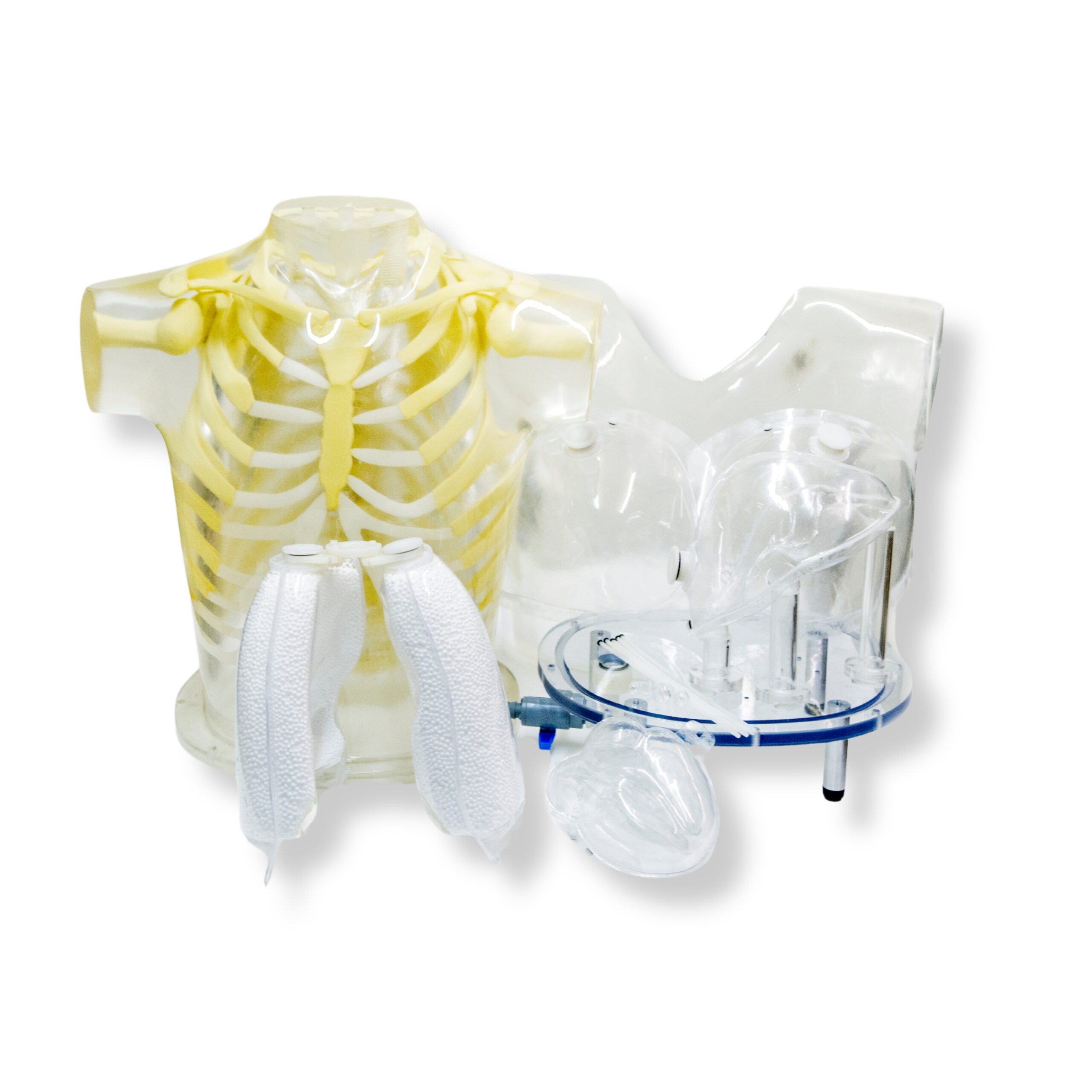
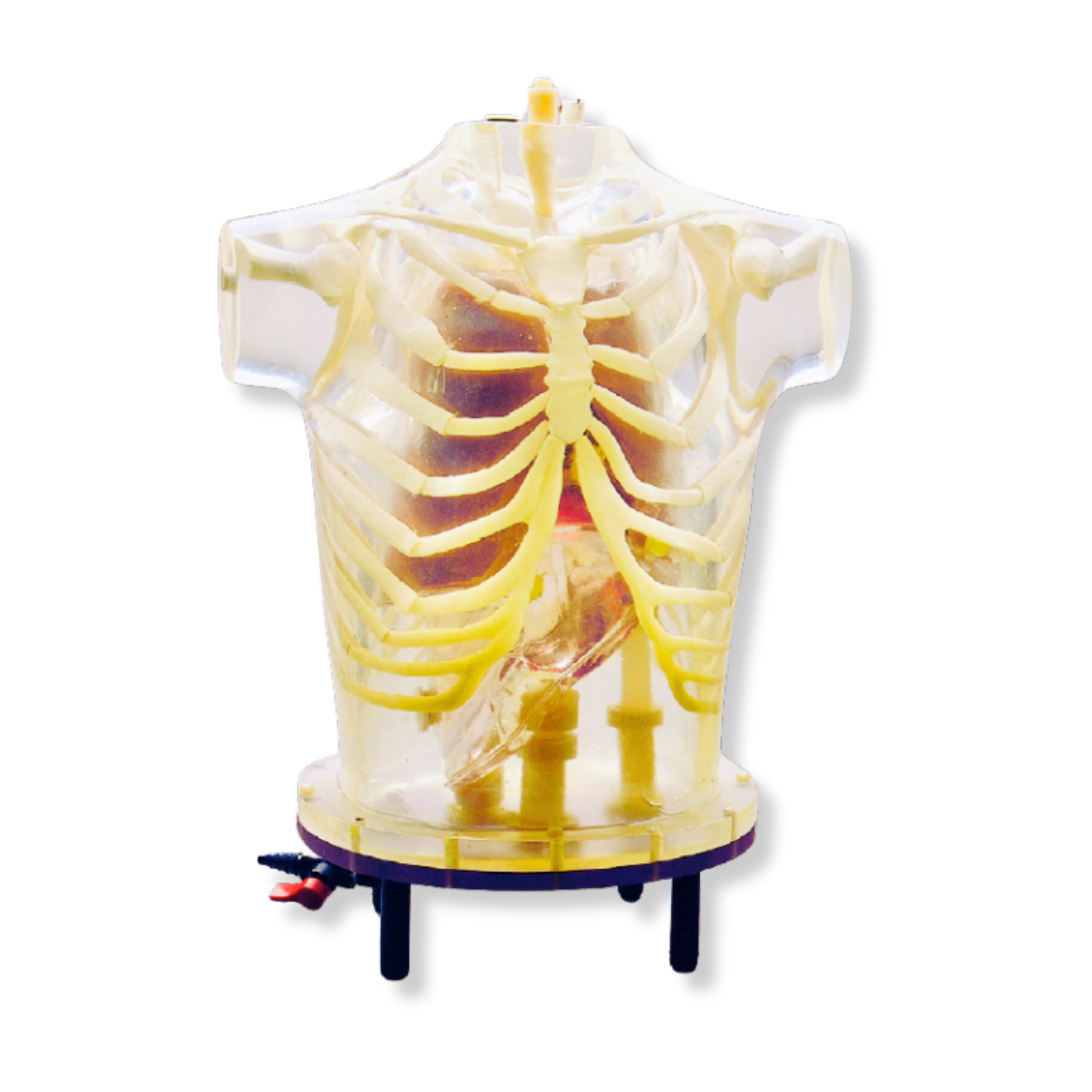
Heart & Thorax Phantom
EVALUATION OF DETECTABILITY, EXTENT, AND SEVERITY OF MYOCARDIAL INFARCTS IN PATIENTS
- Receptor Quantification as a Function of Uptake Ratio(s)
- Partial Volume Effects
- Scatter and Attenuation – Correction Schemes
- Threshold for Changes in Uptake(s)
- Comparison of Different Acquisition Modes, e.g. 2D vs. 3D Pet(s)
RSD’s Heart & Thorax Phantom is designed to provide different reconstruction strategies, imaging protocol for patients, and testing and validation of image registration techniques. It also delivers a valid assessment of mammoscintigraphy techniques.
Myocardial perfusion SPECT is a widely-used, non-invasive method for the diagnosis and management of patients with coronary disease. However, non-uniform photon attenuation, Compton scatter, limited and depth-dependent spatial resolution, as well as image noise, limit the ability of SPECT to obtain images that reliably represent the true tracer distribution. The non-uniform attenuation of the thorax is the most significant factor limiting the diagnostic efficacy of myocardial SPECT.
The currently used attenuation, scatter and resolution correction methods are suboptimal, since they do not provide improvement in the 25% false-negative findings in a group of about 100 patients with luminal diameter stenoses of at least 50%(1). Furthermore, the ability to detect multivessel disease was 70% without and 47% with corrections. This finding implies that myocardial SPECT can seriously underestimate the extent of disease in high-risk patients. On the other hand, the false positive findings in the group with a low probability of coronary disease were reduced from 14% without corrections to 3% with corrections.
Obviously, further improvements in both hardware and software for myocardial SPECT are necessary before this important diagnostic technique can realize its full potential. These improvements must be carefully evaluated on realistic, anthropomorphic phantoms to improve results in clinical practice.
Heart
An accurately anatomic heart model is based on vacuum-formed shells. It was designed using high resolution, contrast-enhanced, ultrafast CT data from a normal patient, slightly modified to facilitate its use.
The left and right chambers are connected at the atrium region to make a single compartment which can be filled and flushed independently using two ports labeled HC (heart chambers). The right ventricle is slightly modified to allow air to escape during filling. The myocardial wall (MW) has two ports, flushing and independent filling. The volume of the heart chambers is 284 ml, while the volume of the myocardial wall is 238 ml, without inserted defects.
The standard model includes three defects with volumes of 8.9, 13.5, and 41.7 ml, respectively. Each of the defects can be filled separately.
Defects of different dimensions can be ordered at no added cost. A disassembled heart is sent on request, so that dimensions of a special set can be established. Note that different defects cannot be retrofitted in the assembled heart.
Basic Thorax
The thorax is molded of polyurethane, modified for tissue equivalence, with a mass density of 1.10 g/cc. The narrow beam linear attenuation coefficient measured at 140 keV (Tc-99m) is 0.160 cm -1.
The skeleton, embedded in the soft tissue, extends from the suprasternal notch down to L2.The RSD materials closely meet the standards of the International Commission on Radiation Units and Measurement (ICRU) Report No. 44 (Tissue Substitutes in Radiation Dosimetry and Measurement, 1989) for both the cortical and spongiosa components of the human skeleton. The mass densities are 1.88 g/cc for cortical bone and 1.16 g/cc for spongiosa. The narrow beam linear attenuation coefficient for the cortical component, measured at 140 keV, is 0.280 cm-1.
The volume of the thoracic cavity, when all organs (heart, lungs, and liver) are inserted, is about 8,200 ml. It is filled from the top, with either an inert or a radioactive solution, to simulate the thoracic background.
A valve is installed at the base for conveniently draining the phantom. The residue on the walls of the cavity can be easily flushed with the fittings provided at the top of the phantom. A second, smaller fitting is also provided as an air-bleed during filling.
Lungs
Perfusable lungs are molded in hollow, vacuum-formed shells, filled with Styrofoam beads. The final mass density of 0.40 g/cc is attained by adding an inert or radioactive solution through a filling port at the top of each lung shell. Extra sets of lungs can also be furnished for work continuity. The volumes of the left and right lung shells are 907 ml and 1,134 ml, respectively.
Liver
A liver with a volume of 980 ml is included to evaluate the effect of its uptake on quantitative myocardial imaging. It is a vacuum-formed shell, mounted on acrylic tubes to minimize artifacts. The liver can be filled with an inert or radioactive solution.
Fillable External Markers
A set of fillable capsules is provided to serve as external markers. Capsules can be filled with a radioactive solution and attached to the external surface of the phantom. The phantom can then be imaged, using SPECT or PET modalities to compare image registration techniques.
Thorax Overlay, Removable Breasts & Breast Tumors
The thoracic phantom without the overlay simulates an average male patient. The overlay, with or without breasts, simulates a large female or a still larger male patient, respectively. It is then possible to evaluate the effect of additional attenuation and scatter on quantitative myocardial imaging.
The volume of each vacuum-formed breast is 972 ml. A tumor, filled with a radioactive solution can be inserted to evaluate the planar and tomographic imaging techniques used for mammoscintigraphy. A set of breast tumors (3, 6, 9, 12, and 15mm diameters) is included. They are supported by thin, threaded nylon rods which pass-through ports and are sealed by O-rings. They can be bent by hand to reach any part of a breast.
The thorax is mounted on a base plate with an O-ring seal. Four rubber feet provide space under the phantom for drain fittings the base plate is removed readily to provide access to the interior of the phantom.
A knob at the top of the neck secures a rod that passes through to the heart/lung subassembly for disassembly.
Model Numbers
| RS-800T | Heart & Thorax Phantom |
| RS-801 | Thoracic Cavity with the bottom plate |
| RS-802 | Perfusable Lungs with Styrofoam beads (pair) |
| RS-804 | Heart with two hollow defects in myocardial wall – standard size or to fit |
| RS-805 | Liver Shell ONLY |
| RS-806 | Chest Overlay |
| RS-807 | Removable Breast with set of tumors |
| RS-809 | Set of 10 threaded nylon tumor support rods |
| RS-810 | Set of 5 external fillable markers |
| RS-811 | Tumors ONLY (with rods sizes 3, 6, 9, 12, or 15mm) |
Applications
- Myocardial perfusion SPECT image quality evaluation
- Attenuation corrections
- Evaluating patient doses
- Training for SPECT/PET imaging
- Quantitative phantom for SUV validation
Modalities
- SPECT/PET
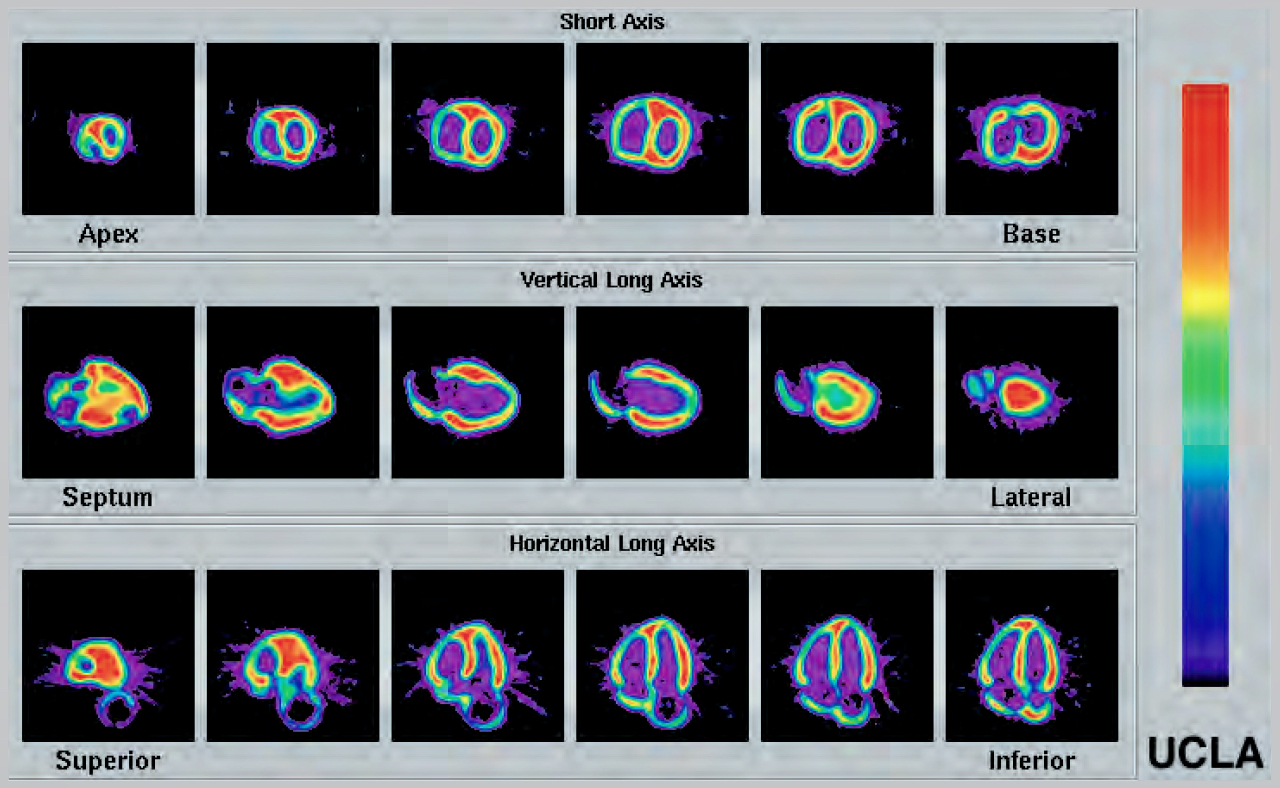
Clinical Images
Publication References
Gallivanone F, Carne I, Interlenghi M, et al. A Method for Manufacturing Oncological Phantoms for the Quantification of 18F-FDG PET and DW-MRI Studies. Contrast Media Mol Imaging. 2017;2017:3461684. Published 2017 Sep 7. DOI: 10.1155/2017/3461684
Heikkinen JO, Kuikka JT, Rautio PJ. Interdepartmental Audit with an Anatomically Realistic Lung Phantom. Journal of Nuclear Medicine Technology. 2006 Mar;34(1):34-42. PMID: 16517967
Doshi N, Basic M, Cherry S. Evaluation of the Detectability of Breast Cancer Lesions Using a Modified Anthropomorphic Phantom. Journal of Nuclear Medicine, 39, 1951-7. 1998. PMID: 9829588