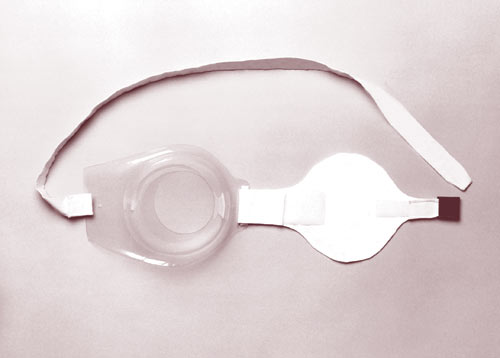
The Treatment Brassiere
Model Numbers
| Model Number | Product Description | Chest Breadth | Specify Breast Volume |
| BR-100-TBC | Basic Brassiere Library with Caddy, 40 different cups | All | All |
| BR-100 | Basic Brassiere Library without Caddy, 40 different cups | All | All |
| BR-100S | Small Group | 27-32 cm | 200 300 400 500 600 |
| BR-100M | Medium Group | 32-36 cm | 250 500 750 1,000 1,250 |
| BR-100L | Large Group | 36-42 cm | 500 750 1,000 1,250 1,500 |
| BR-100XL | Extra Large Group | Over 42 cm | 1,000 1,500 2,000 2,500 3,000 |
Applications
- Simplified application of IMRT to breast treatments
- Accurately repeats both shape and position, treatment after treatment
- Reduces dose to the lungs, heart and ribs
Modalities
- Intensity-Modulated Radiation Therapy (IMRT)
Data on increased skin dose due to the ipsilateral cup were submitted to the FDA. These data provided a comparison of skin dose increased by the Treatment Brassiere with the increase found for various thermoplastic positioning devices on the market. The skin dose due to the cup material (0.50 mm thick for small cups to about 0.63 mm thick for very large cups) was found to be substantially less than for other positioning devices. The FDA has issued a 510 (k) Marketing Clearance for the Treatment Brassiere.
Cup Selection
| Model Number | Product Description | Chest Breadth | Specify Breast Volume |
| BR-100-TBC | Basic Brassiere Library with Caddy, 40 different cups | All | All |
| BR-100 | Basic Brassiere Library without Caddy, 40 different cups | All | All |
| BR-100S | Small Group | 27-32 cm | 200 300 400 500 600 |
| BR-100M | Medium Group | 32-36 cm | 250 500 750 1,000 1,250 |
| BR-100L | Large Group | 36-42 cm | 500 750 1,000 1,250 1,500 |
| BR-100XL | Extra Large Group | Over 42 cm | 1,000 1,500 2,000 2,500 3,000 |
Cup selection begins by measuring a patient’s chest breadth when supine and wearing her clothing brassiere. This indicates the group containing the right cup for her. The cups of the group (five each for left or right) are removed from the Treatment Brassiere Caddy and the cup making the best fit is selected. (Clothing Brassiere sizes cannot be used for cup selection because of poor correlation between clothing brassiere sizes and breast volumes.)
| Clothing Cup Size* | Observed Volume |
| A | 94 - 225 ml |
| B | 146 - 458 ml |
| C | 267 - 835 ml |
| D | 499 – 1,120 ml |
| DD | 988 – 1,427+ ml |
| DDD | 2,765 – 3,176 ml |
These data are based on 75 models. Some cup volumes shown are altered from other cups as required for continuous gradation.
*As reported by models.